|
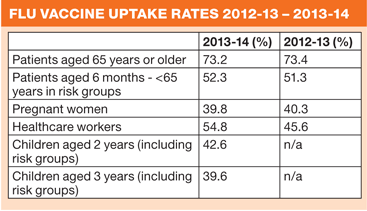
May 2014 InfluenzaAmbitious targets set for next winter flu campaignPractice nurses have been urged to start planning now for the 2014-15 flu immunisation programme to meet an ambitious uptake target of at least 75% for all those in clinical risk groups – not forgetting practice staff. NHS England has written to general practices thanking ‘everyone involved for all their hard work’ over the 2013-14 campaign, but says that the overall uptake last winter fell ‘below the 75% aspiration, and this remains a priority’. ‘This year we are asking that GP practices prioritise improvements in vaccine uptake over and above last season in those with chronic liver and neurological disease, including people with learning disabilities, who are at the highest risk of mortality from flu but have the lowest rate of vaccine uptake.’ The other priority group should be pregnant women, NHS England says. Last winter saw low levels of circulating flu, but the seasonal flu letter says ‘we must not be complacent.’ During the previous winter flu season, an estimated 11,000 deaths were attributable to flu, the highest number since 2008-09. NHS England has urged practices to start vaccinations as early as possible between September and early November before flu starts circulating in the community, and this year practices will have the opportunity to count vaccinations given even earlier, in August, towards their QOF targets. This year’s target groups are: All children aged 2 - <5 years on 1 September 2014, and All patients aged 65 years and older, and children aged 6 months or older who have:
- COPD - Children who have previously been admitted to hospital for lower respiratory tract disease
And:
The programme to vaccinate children has been extended for this year to include all children aged 2, 3 and 4 years old, but not 5 years or older on 1 September 2014. The vaccine for children is also changing, from the trivalent live attenuated influenza vaccine (LAIV) used last year to a quadrivalent LAIV (Fluenz Tetra®). Most children should be offered a single dose, but those in clinical at-risk groups between the age of 2 and 9 years should be given two doses, at least four weeks apart. VACCINE VIRUS STRAINSFlu viruses change continuously and the World Health Organization (WHO) monitors the epidemiology of flu viruses throughout the world. Each year it makes recommendations about the strains to be included in vaccines for the forthcoming winter: www.who.int/influenza/vaccines/virus/recommendations/2014_15_north/en/ It is recommended that trivalent vaccines for use in the 2014-2015 influenza season (northern hemisphere winter) contain the following:
WHO says quadrivalent vaccines containing two influenza B viruses should contain the above three viruses and a B/Brisbane/60/2008-like virus. Source: The National Flu Immunisation Programme 2014-15 https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/306646/FluImmunisationLetter2014_ accessible.pdf |
 |
Back to top